Deep Ensemble learning and quantum machine learning approach for Alzheimer’s disease detection

The focus of this work is the development of an ensemble DL-based quantum machine learning classification model for the diagnosis of AD disease. Deep learning architectures are the most widely employed for processing and analyzing brain images in research projects8,18. In this paper, we introduced a method based on ensemble learning and quantum machine learning classification algorithms that analyze MRI brain images and extract meaningful features for successful classification of AD stages. According to Fig. 1, The proposed algorithm uses sequential steps, The ADNI1 and ADNI2 MRI image data sets are first prepared, pre-processed, and then combined to use in the proposed approach. This is followed by the building of an ensemble model, and its parameters are configured for feature extraction and the features are fed into QSVM for classification of AD dementia stages. Finally, the performance of the proposed model is conducted and compared against the other cutting-edge methods. To judge the models, numerous performance metrics, including accuracy, recall, precision, F1- score, and AUC were calculated. The results implied that the ensemble model-based QSVM is superior to the other cutting-edge methods in terms of performance. Likewise, it can be inferred that effective outcomes can be achieved by combining quantum classifiers and ensemble learning.
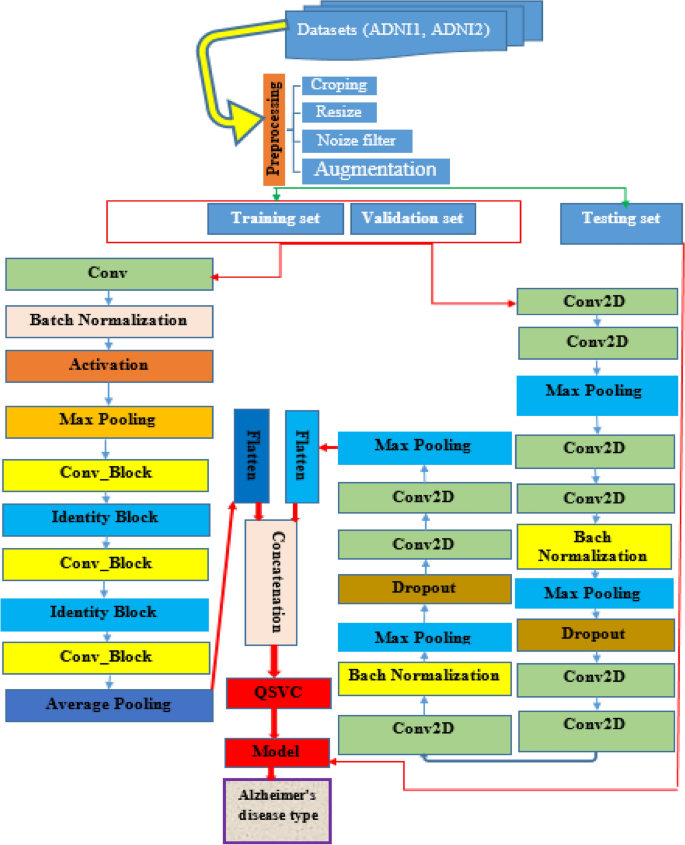
Architecture of the proposed ensemble model with QSVC/QSVM.
As visualized in Fig. 1, the VGGNet model consists nine convolutions, two batch normalizations, three max-pooling, two dropouts, and one flattened layer. A dropout layer is inserted below each max pooling and dense layer to overcome overfitting. while the ResNet model consists of one convolution, one max-pooling, one average polling, one batch normalization, one activation, two identity blocks, three conv blocks, and one flattened layer. Finally, the extracted and flattened features from the two models are concatenated to classify into four Alzheimer’s disease types using the QSVM classifier.
Data and material
The data used to develop the model in our work was retrieved from different sources, such as the ADNI 1 dataset and the ADNI2 dataset from the Kaggle databases (ADNI_Extracted_Axial (kaggle.com)). Alzheimer’s disease Neuroimaging Initiative is a large-scale study focusing on the early detection and progression monitoring of AD. We combine ADNI 1 and ADNI 2 datasets of AD since both are obtained from MRI scans of AD patients in different time stamps. Combining different sources of datasets used to boost deep learning training algorithms for performance improvement. We used only MRI scans of patients from both sources to increase the number of datasets. The high number of datasets helps to reduce overfitting problems in DL algorithms. The summary of our dataset is in Table 1.
Preprocessing
The chosen data was pre-processed using a standard processing pipeline. We used a cropping algorithm to eliminate the bone and skeleton portions of the MRI images since this superfluous portion is not significant for AD classification. The original dataset had an image resolution of 176*208. We must scale the MRI image to 128*128 pixels in width and height due to hardware limitations. We lowered the dataset’s dimensionality to 5 qubits in order to use it for quantum classification. In our study, we used an adaptive median filter to remove outliers for the facilitation of a reliable classification process. The augmentation technique was applied to increase the number of the Alzheimer’s disease dataset. The efficacy of the augmentation in terms of model over fitting was also improved to increase the generalization capability of deep learning models. Also, there is a class imbalance problem so to alleviate that we used data augmentation comprising arbitrary height and width shift (range, 0–10%) and zooming (range, 0–8%) on the training set.
Deep learning
Deep learning models, specifically convolutional neural networks (CNN), have revolutionized disease detection in healthcare. We employed the prepared VGG-16 convolutional neural network model, which was enhanced by freezing some of the layers. Simonyan and Zisserman identified the 16-layer convolutional architecture referred to as the VGG-16 model in 2014. The VGG-16 model pertains itself a large network with about 138 million parameters. It piles many convolutional layers to construct deep neural networks that boost their ability to learn hidden features. The network’s input image possesses dimensions of (224 × 224 × 3). It additionally includes 16 convolutional layers that work as a fixed size filter (3 × 3) and 5 layers of Max grouping that encompass the entire network in size (2 × 2)21. ResNet 50 is perhaps the most powerful convolutional neural network architecture available in the recent decade22. It was also selected as the winner of the ILSVRC competition. ResNet-50, a convolutional neural network with 50 layers, is one of the versions of ResNet. A total of 48 convolution layers are included 1 Max pooling and 1 Average pooling layer. It is a deep residual learning framework built on a neural network. It can resolve the vanishing gradient problem even when working with incredibly dense neural networks. ResNet 50, despite the fact it contains 50 layers, has around 23 million trainable parameters, which are significantly less than the trainable parameters of previous architectures. In the residual network rather than learning features, it learns residuals which are the subtraction of learned features from the layer inputs ResNet connects the input of the nth layer directly to an (n + x) th layer, allowing additional layers to be stacked and a deep network established.
The proposed ensemble model for feature extraction
The performance of categorizing biomedical signals is enhanced by feature extraction. To increase the efficacy of the classifier, feature extraction intends to discover the most relevant and valuable set of features (unique properties). The most important step in classifying biomedical signals is feature extraction since improperly chosen features could cause the classification performance to suffer12. Unlike traditional methods that are time-consuming and require specialized knowledge for feature extraction, deep learning can automatically extract relevant features from input images, resulting in improved prediction accuracy23. In this research, we used VGG16- ResNet50 as a base model to exploit the local spatial characteristics of the images. A multitude of features is retrieved from pre-processed image data. The features of MRI images of AD patients are explored by ensemble deep learning models namely customized version of VGG16-ResNet50. Since different CNN architectures can capture diverse information of input images, which increase performance than a single model, the concatenation process of two model features integrates the information from different CNNs to create a more discriminative feature representation than using the feature extracted from a single CNN model24. In our Ensemble model, we concatenated the features obtained from the two models as shown in the architecture depicted in Fig. 1.
As previously discussed, the proposed model aims to accurately diagnose AD disease by concatenating deep features extracted from MRI images by using two different models (customized CNN architecture of VGGNet and ResNet50). First, a VGGNet model is proposed to extract features from MRI images. Correspondingly, the ResNet model extracts features from the same images. To end with, the extracted features from these models are flattened and concatenated into a single classification descriptor. Then, the extracted features are fed into the QSVM classifier.
Classification with quantum machine learning
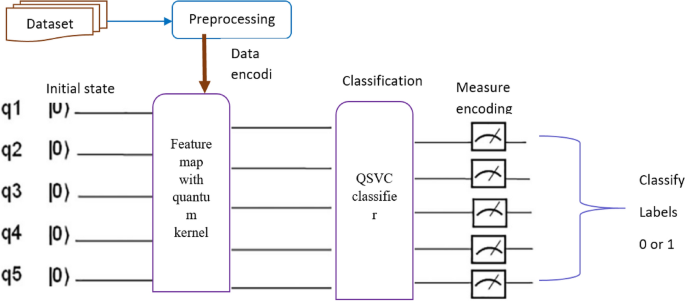
Support vector machine is a classical machine learning algorithm that uses training data from the sets to classify vectors in a feature space into one of two sets. It attempted to discover a high-probability optimum separation hyperplane between two distinct groups or class in a set of samples, with whole training samples of the class located on one side of the hyperplane. The linear discrimination problem is to develop a hyperplane that may be used as a decision-boundary classification task while also providing substantial differences between two class regions25. Quantum-enhanced machine learning techniques can accomplish several tasks, among them lowering training time, managing complex network topology, automatically modifying network hyper parameters, performing complex matrix and tensor manipulation at high speeds, and using quantum tunneling to achieve actual objective function goals, in contrast to traditional machine learning algorithms26. In quantum computers, Quantum SVM is the quantum counterpart of the classical SVM. The QSVM has a quantum advantage over the classical SVM in situations where it is challenging to estimate the feature map classically. Using a quantum kernel in QSVM algorithms, quantum computers can accelerate learning by using a quantum kernel11. Using quantum feature maps that map data points to quantum states, classical data can be encoded to be processed by a quantum computer10. In the following direction, QSVM, a quantum machine learning algorithm was adopted to take the essential features obtained by the ensemble model and classify the MRI scan as AD stages.
Figure 2 illustrates the structure of the QSVM algorithm, in which the feature maps are flattened by applying the dense layer. The flattened feature maps were subsequently mapped to quantum spaces using a 5-qubit feature map. By taking the inner product of the quantum feature maps, the quantum kernel maps the quantum state data points into higher-dimensional space. Following the QSVM classifier fitting to the training data and evaluating the performance of the model using the test data, for each classical input, the measurements decode the quantum data into the corresponding classical output data.
link